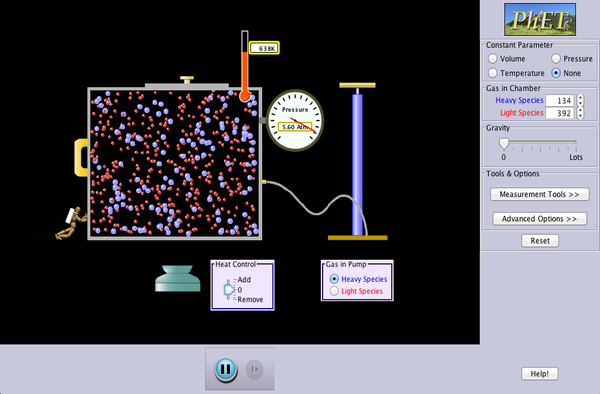
Paul Anderson helps us out again. This video has a great set of demonstrations using a set of interactive animations. Students are expected to complete a worksheet based on this video:
The simulation that Mr. Anderson uses in the video can be accessed online, at the University of Colorado, HERE:
Thursday, January 25, 2018
Friday, January 12, 2018
POWER POINT: PRESSURE AND TEMPERATURE CONVERSIONS
Students: here is the (brief) Power Point containing information about the physical basis of temperature and pressure in the metric system, and how to convert units related to the same:
VIDEO: TEMPERATURE
As we begin our notes on the 'Kinetic Theory of Gases', it is important to realize that the random, dynamic motion of a very large set of tiny (sub-MICROSCOPIC) particles produces the phenomena we can observe at our own, relatively large (MACROSCOPIC) scale, like pressure and temperature.
Paul Anderson demonstrates this in the following video. Students will receive an accompanying worksheet based upon it!
Paul Anderson demonstrates this in the following video. Students will receive an accompanying worksheet based upon it!
Sunday, January 7, 2018
SPRING 2018 SEMESTER BEGINS!
WELCOME BACK!
Students in Mr. Hatfield's Chemistry classes will be spending much of the first week of the Spring 2018 semester completing a four-part packet of handouts which reviews math routines taught and rehearsed in the Fall semester.
On ATLAS, this packet will display as four separate classwork assignments: Molar Mass, Two-Step Problems, Three-Step Problems and Stoichiometry 1. Each is worth 20 points, but they are shown on the Syllabus below as a single (80-point) assignment:
Students should've also begun answering the Section Assessment Questions from Chapter 13, and beginning on Tuesday of the second week, begun to absorb new material on kinetic molecular theory and gas laws.
Students in Mr. Hatfield's Chemistry classes will be spending much of the first week of the Spring 2018 semester completing a four-part packet of handouts which reviews math routines taught and rehearsed in the Fall semester.
On ATLAS, this packet will display as four separate classwork assignments: Molar Mass, Two-Step Problems, Three-Step Problems and Stoichiometry 1. Each is worth 20 points, but they are shown on the Syllabus below as a single (80-point) assignment:
Subscribe to:
Comments (Atom)